Carbon electron configuration number6/21/2023  In any atom with two or more electrons, the repulsion between the electrons makes energies of subshells with different values of l differ so that the energy of the orbitals increases within a shell in the order s < p < d < f. The energy of atomic orbitals increases as the principal quantum number, n, increases. The specific arrangement of electrons in orbitals of an atom determines many of the chemical properties of that atom. This allows us to determine which orbitals are occupied by electrons in each atom. Having introduced the basics of atomic structure and quantum mechanics, we can use our understanding of quantum numbers to determine how atomic orbitals relate to one another. Relate electron configurations to element classifications in the periodic table.Identify and explain exceptions to predicted electron configurations for atoms and ions.Derive the predicted ground-state electron configurations of atoms.This is the reason that carbon is different from the silicon and both of them are different from other elements of this group.By the end of this section, you will be able to: The penultimate shell of carbon contains the s 2 electrons, silicon has s 2p 6 electrons and germanium contains the s 2p 6d 10 electrons and is unsaturated. So, they have an electronic configuration of s 2p 2 in their valance shell. Two of the electrons are in the s orbital and the remaining two are in the p orbital. The electronic configuration of group 14 elements shows that these elements have four electrons in their valance (ultimate shell). 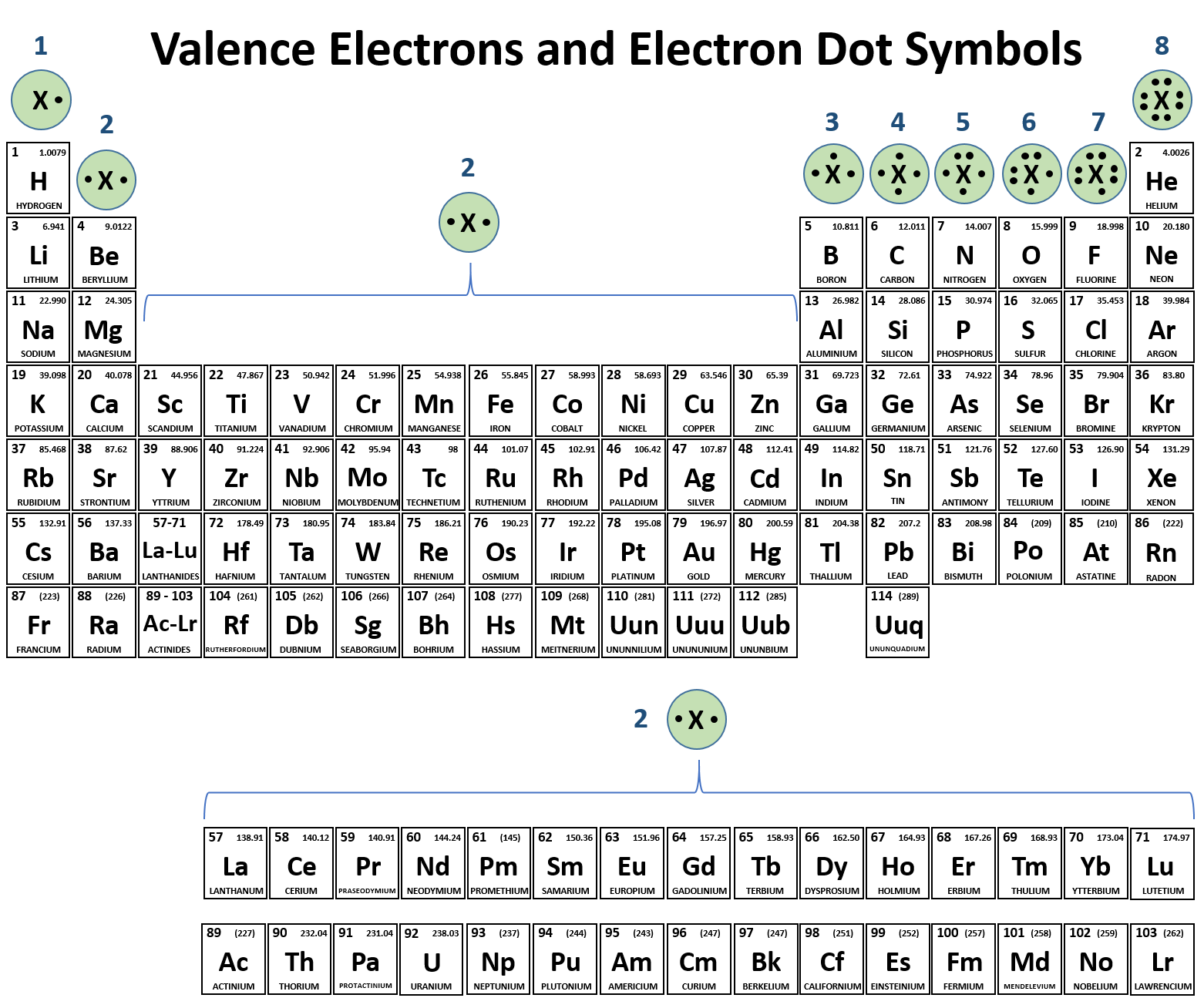
The electronic configuration for each element of this group is given below. There are two electrons in the outermost p orbitals of these elements. The general electronic configuration of group 14 elements is ns 2np 2. It is due to the fact that orbitals specify the position and shape of the regions of the space that is occupied by the electrons. Orbitals and electrons are very closely related however, orbitals provide a much accurate picture of the electronic configuration of an atom. Each column in the periodic table reflects the number of the electrons in the valance shells of each of the element which in turn strongly determine how the element will react. Typically, the sharing of electrons is observed to complete the valance shells by forming the multiple bonds with the other atoms. Carbon group the group 14 elements have four electrons in the outer shell. In neutral atoms, the number of electrons is equal to the number of the protons and atomic number can be easily determined by using the electron number. In the periodic table, elements are placed according to their atomic numbers that how many protons they have. Hund’s rule states that the pairing of electrons in the orbital only takes place when there is one electron in each subshell. 
The Pauli exclusion principle states that all four quantum numbers for any of the two electrons in an atom can never be the same. Another rule is defined by the Pauli and he defined a set of the unique quantum numbers for each of the electron. So, electrons fill the energy levels as defined by Aufbau’s principle. 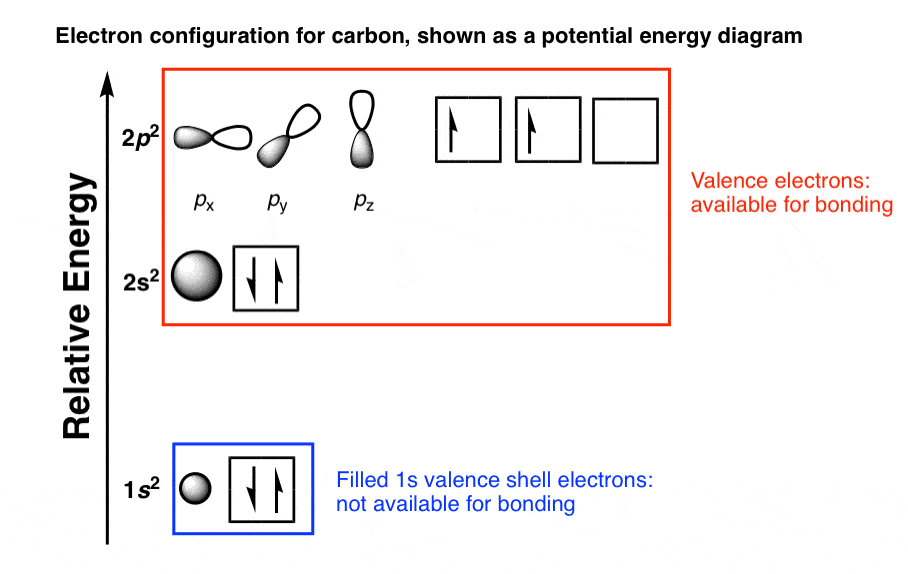
Electrons fill the orbitals of the atom in such a way that the energy of the atom is at the minimum level. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
